Lateral Flow Assays: Principles, Designs and Reagents

Lateral Flow Tests
Lateral flow assay development is the successful production of a simple to use diagnostic test for validating the presence or absence of a wide range of pathogens, biomarkers and antibodies in a specific species. Rapid lateral flow tests are used for qualitative and quantitative analysis. Qualitative by visually indicating if a substance is present and quantitative by determining how much of that substance is present. Lateral flow immunoassay tests are highly important and versatile within point-of-care diagnostics.
How Do Lateral Flows Work?
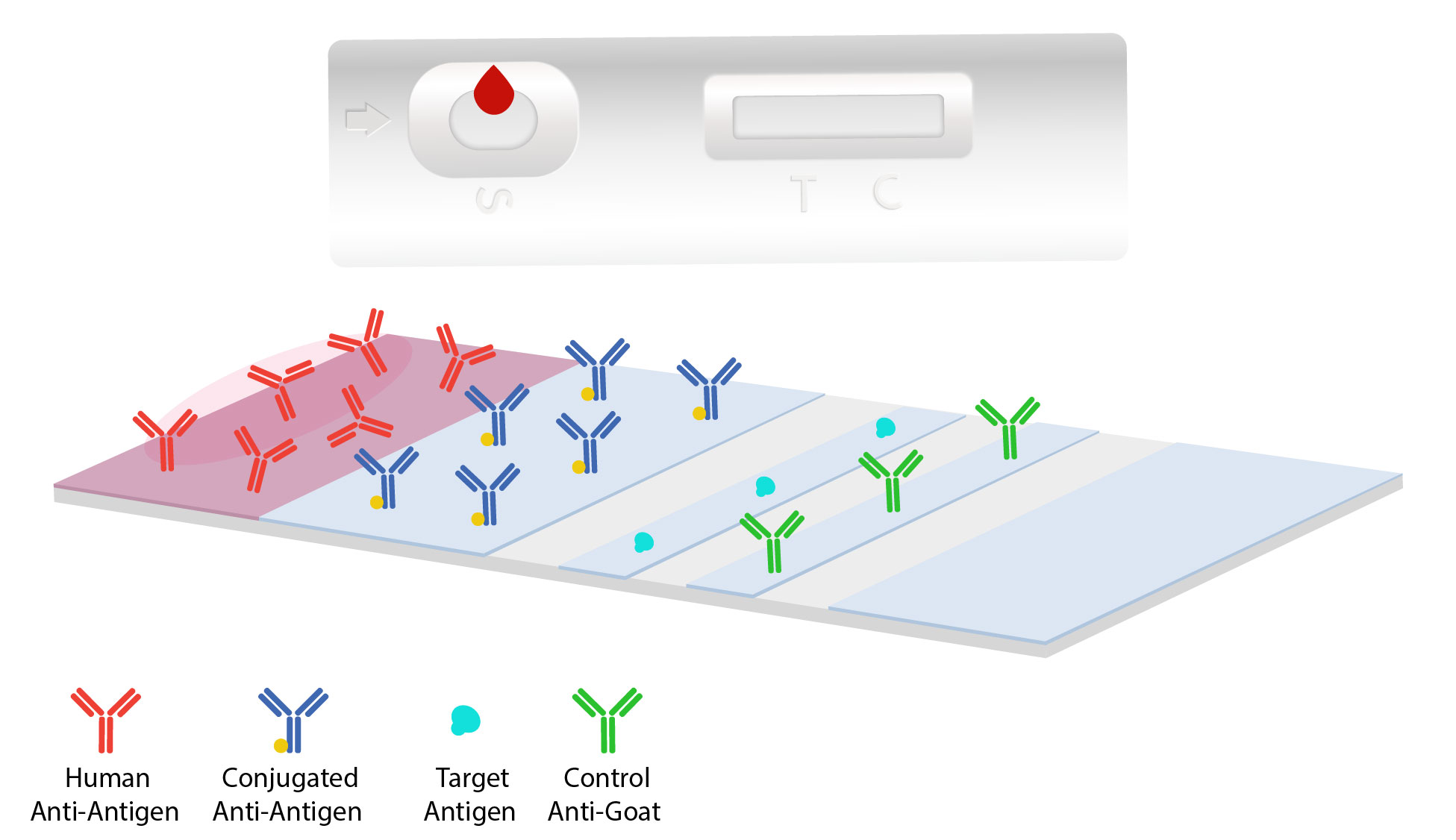
Components of the test flow laterally through the devices grid allowing for antigens to combine with antibodies to signal a response between the complexes. The components involved in a lateral flow assay are the sample pad, a conjugate pad, a nitrocellulose membrane, and a absorbent pad.
Subjects specimen is collected and introduced to the sample pad or device.
Immunoassay Development Services with Leinco
Our experienced research and scientific team assists in rapid point-of-care lateral flow assay development from the planning to validation phase. Leinco provides high quality assay development services and products while meeting rigorous deliverables and specifications. Antibodies used in lateral flow tests should have high sensitivity, specificity, purity, and stability to accomplish performance requirements. We work closely with customers throughout every step of their assay development process, assuring our components are optimized for your IVD diagnostic and research needs.